Japan (EN)
Select your region or country.
ORCA-Fusion Digital CMOS camera
C14440-20UP
The ORCA®-Fusion, built from the sensor up, balances the complex nuances of camera features to provide beautiful images and robust data at all lights levels, but especially in tough low-light conditions. The exceptionally low and highly uniform read noise of the ORCA-Fusion means that when the sample emits even just a handful of photons, either by default or by experimental design, they are not lost in the noise, but detected and reliably quantified. After all, when you want to hear a whisper it’s best to be in a quiet place.
ORCA is a registered trademark of Hamamatsu Photonics K.K. (China, EU, France, Germany, Japan, UK, USA).
Features
- High resolution : 2304 × 2304 (5.3 Megapixels)
- High speed : 100 frames/s (at 2304 × 2048 ROI)
- Dynamic range : 21,400:1
- Pixel size : 6.5 μm × 6.5 μm
- Read noise : 0.7 electrons, rms (Ultra quiet Scan)
- Prnu : 0.06 % (@ 7500 electrons)
- DSNU : 0.06 electrons
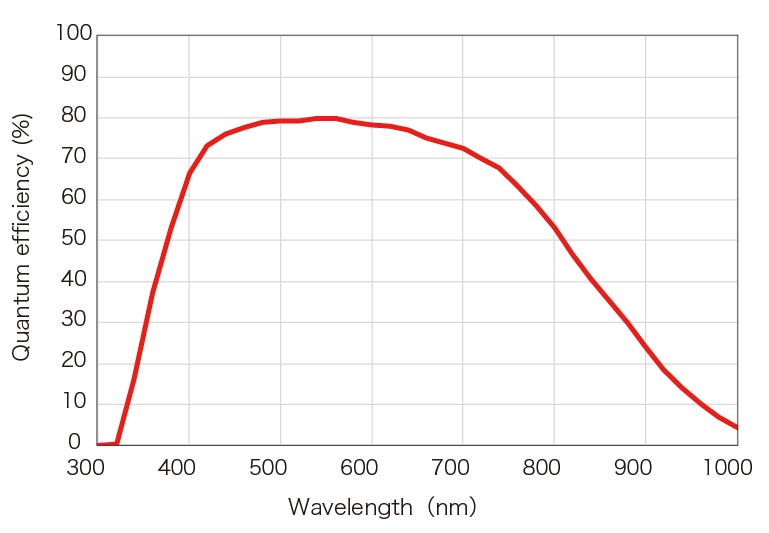
- Peak QE : 80 %
- Patented Tools for Advanced Imaging: Lightsheet Readout Mode
Lightsheet Readout Mode [Patented]
“Lightsheet Readout Mode” is a unique and patented feature of Hamamatsu sCMOS cameras which can improve signal to noise ratios in Lightsheet microscopy.
For more information about the principle and features of Lightsheet Readout Mode, please see details from below.
Publications
ORCA-Fusion
| Authors | Title | Source |
|---|---|---|
| Piatkevich, K.D., Bensussen, S., Tseng, Ha. et al. | Population imaging of neural activity in awake behaving mice | Nature 574, 413–417 (2019). |
| Silvia Benito-Kwiecinski, Stefano L. Giandomenico, Magdalena Sutcliffe, Erlend S. Riis, Paula Freire-Pritchett, Iva Kelava, Stephanie Wunderlich, Ulrich Martin, Gregory A. Wray, Kate McDole, Madeline A. Lancaster | An early cell shape transition drives evolutionary expansion of the human forebrain | Cell, Volume 184, Issue 8, 2021, Pages 2084-2102.e19, |
| Madhav Mantri, Gaetano J. Scuderi, Roozbeh Abedini-Nassab, Michael F. Z. Wang, David McKellar, Hao Shi, Benjamin Grodner, Jonathan T. Butcher & Iwijn De Vlaminck | Spatiotemporal single-cell RNA sequencing of developing chicken hearts identifies interplay between cellular differentiation and morphogenesis | Nat Commun 12, 1771 (2021). |
| Kiryl D. Piatkevich, Seth Bensussen, Hua-an Tseng, Sanaya N. Shroff, Violeta Gisselle Lopez-Huerta, Demian Park, Erica E. Jung, Or A. Shemesh, Christoph Straub, Howard J. Gritton, Michael F. Romano, Emma Costa, Bernardo L. Sabatini, Zhanyan Fu, Edward S. Boyden & Xue Han | Volumetric live cell imaging with three-dimensional parallelized RESOLFT microscopy | Nat Biotechnol 39, 609–618 (2021). |
| Xinyu Wang, Chong Liu, Siwei Zhang, Huiwen Yan, Liwen Zhang, Amin Jiang, Yong Liu, Yun Feng, Di Li, Yuting Guo, Xinyao Hu, Yajing Lin, Pengcheng Bu, Dong Li, | N6-methyladenosine modification of MALAT1 promotes metastasis via reshaping nuclear speckles | Developmental Cell, Volume 56, Issue 5, 2021, Pages 702-715.e8, |
| Emily C. Wheeler, Anthony Q. Vu, Jaclyn M. Einstein, Matthew DiSalvo, Noorsher Ahmed, Eric L. Van Nostrand, Alexander A. Shishkin, Wenhao Jin, Nancy L. Allbritton & Gene W. Yeo | Pooled CRISPR screens with imaging on microraft arrays reveals stress granule-regulatory factors | Nature Methods volume 17, pages636–642 (2020) |
| Scott M. Emrich, Ryan E. Yoast, Ping Xin, Vikas Arige, Larry E. Wagner, Nadine Hempel, Donald L. Gill, James Sneyd, David I. Yule, Mohamed Trebak, | Omnitemporal choreographies of all five STIM/Orai and IP3Rs underlie the complexity of mammalian Ca2+ signaling | Cell Reports, Volume 34, Issue 9, 2021, 108760, |
| Tomomi Suzuki, Tetsuo Mioka, Kazuma Tanaka & Akira Nagatani | An optogenetic system to control membrane phospholipid asymmetry through flippase activation in budding yeast | Sci Rep 10, 12474 (2020). |
| Nicholas S. Groves,Merissa M. Bruns and Schuyler B. van Engelenburg | A Quantitative Live-Cell Superresolution Imaging Framework for Measuring the Mobility of Single Molecules at Sites of Virus Assembly | Pathogens 2020, 9(11), 972 |
| Pierre Stömmer, Henrik Kiefer, Enzo Kopperger, Maximilian N. Honemann, Massimo Kube, Friedrich C. Simmel, Roland R. Netz & Hendrik Dietz | A synthetic tubular molecular transport system | Nat Commun 12, 4393 (2021). |
| Hugh Sparks, Lucas Dent, Chris Bakal, Axel Behrens, Guillaume Salbreux, and Chris Dunsby | Dual-view oblique plane microscopy (dOPM) | Biomed. Opt. Express 11, 7204-7220 (2020) |
| Philipp Zelger, Lisa Bodner, Martin Offterdinger, Lukas Velas, Gerhard J. Schütz, and Alexander Jesacher | Three-dimensional single molecule localization close to the coverslip: a comparison of methods exploiting supercritical angle fluorescence | Biomed. Opt. Express 12, 802-822 (2021) |
PC recommendations
With the introduction of the ORCA-Fusion, users are now able to stream 5 megapixel images to their computers 89 frames per second. The computer recommendations for this high data rate can be met by using the guidelines listed in the PC Recommendations for ORCA-Fusion.
Software

Our software provides the interface to access all our carefully engineered camera features, from simply setting exposure to orchestrating complex triggering for multidimensional experiments.
Specifications
| Type number | C14440-20UP |
|---|---|
| Imaging device | sCMOS |
| Effective no. of pixels | 2304(H)×2304(V) |
| Cell size | 6.5 μm(H)×6.5 μm(V) |
| Effective area | 14.976 mm (H)×14.976 mm (V) |
| Full well capacity | 15 000 electrons *1 |
| Readout speed | Fast scan: 11.22 ms (89.1 frames/s with CoaXPress or 31.6 frames/s with USB 3.0) (2304 x 2304) *5 Standard scan: 42.99 ms (23.2 frames/s with CoaXPress or USB 3.0) (2304 x 2304) *5 Ultra quiet scan: 184.4 ms (5.4 frames/s with CoaXPress or USB 3.0) (2304 x 2304) *5 |
| Readout noise | Fast scan:1.4 electrons rms *1 Standard scan:1.0 electrons rms *1 Ultra quiet scan:0.7 electrons rms *1 |
| Exposure time | Fast scan: 17 μs to 10 s (4.87 μs step) Standard scan: 65 μs to 10 s (18.65 μs step) Ultra quiet scan: 280 μs| 10 s (80.00 μs step) |
| Cooling method | Peltier cooling |
| Cooling temperature | Forced-air cooled: -5 ˚C (Ambient temperature: +25 ˚C) Water cooled: -5 ˚C (Water temperature: +25 ˚C) Water cooled (Max cooling): Less than -15 ˚C *1,*3 |
| Dark current | Cooling temperature(-5 ˚C): 0.5 electrons/pixel/s *1,*4 Cooling temperature(-15 ˚C): 0.2 electrons/pixel/s *1,*4 |
| Dynamic range | 21 400 : 1 *1,*2 |
| External trigger mode | Edge, Level, Sync readout, Start, Global reset edge, Global reset level |
| External trigger signal routing | SMA |
| Trigger delay function | 0 s to 10 s in 1 μs steps |
| Trigger output | 3 programmable timing, Global exposure timing, trigger ready, low, high |
| External signal output routing | SMA |
| Interface | CoaXPress (Dual CXP-6) and USB 3.0 *6 |
| A/D converter | 16 bit / 12 bit / 8 bit |
| Lens mount | C-mount |
| Power supply | Approx. 150 VA *7 |
| Ambient operating temperature | 0 ˚C to + 40 ˚C |
| Ambient operating humidity | 30 % to 80 %, with no condensation |
*1 Typical value
*2 Calculated from the ratio of the full well capacity and the readout noise
*3 The water temperature is +20 ˚C and the ambient temperature is +20 ˚C
*4 Dark current depends on cooling temperature
*5 Valid to 4 digits and rounded up to 5th digit
*6 USB 3.1 Gen 1 compatible
*7 The value with AC 240 V. (Approx. 70 VA with AC 100 V)
Spectral response
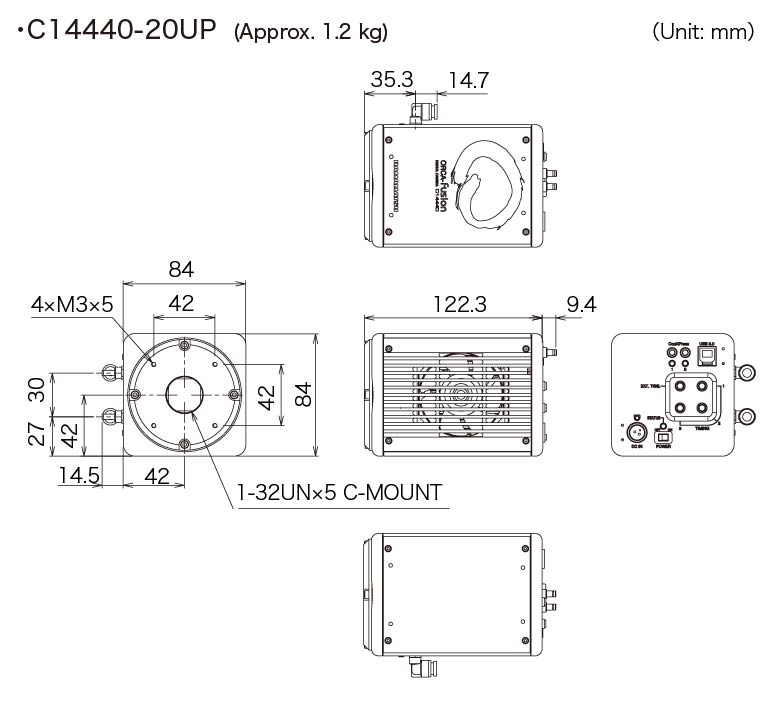
Dimensions
Instruction manual
Special site
This site provides information on scientific cameras.
Since there is a wide range of camera types and performance, it is important to select the best camera for each application.
It introduces technical information, simulation tools, and examples of actual applications to help you fully understand the performance of the camera and select the best one for your application.
- Confirmation
-
It looks like you're in the . If this is not your location, please select the correct region or country below.
You're headed to Hamamatsu Photonics website for JP (English). If you want to view an other country's site, the optimized information will be provided by selecting options below.
In order to use this website comfortably, we use cookies. For cookie details please see our cookie policy.
- Cookie Policy
-
This website or its third-party tools use cookies, which are necessary to its functioning and required to achieve the purposes illustrated in this cookie policy. By closing the cookie warning banner, scrolling the page, clicking a link or continuing to browse otherwise, you agree to the use of cookies.
Hamamatsu uses cookies in order to enhance your experience on our website and ensure that our website functions.
You can visit this page at any time to learn more about cookies, get the most up to date information on how we use cookies and manage your cookie settings. We will not use cookies for any purpose other than the ones stated, but please note that we reserve the right to update our cookies.
1. What are cookies?
For modern websites to work according to visitor’s expectations, they need to collect certain basic information about visitors. To do this, a site will create small text files which are placed on visitor’s devices (computer or mobile) - these files are known as cookies when you access a website. Cookies are used in order to make websites function and work efficiently. Cookies are uniquely assigned to each visitor and can only be read by a web server in the domain that issued the cookie to the visitor. Cookies cannot be used to run programs or deliver viruses to a visitor’s device.
Cookies do various jobs which make the visitor’s experience of the internet much smoother and more interactive. For instance, cookies are used to remember the visitor’s preferences on sites they visit often, to remember language preference and to help navigate between pages more efficiently. Much, though not all, of the data collected is anonymous, though some of it is designed to detect browsing patterns and approximate geographical location to improve the visitor experience.
Certain type of cookies may require the data subject’s consent before storing them on the computer.
2. What are the different types of cookies?
This website uses two types of cookies:
- First party cookies. For our website, the first party cookies are controlled and maintained by Hamamatsu. No other parties have access to these cookies.
- Third party cookies. These cookies are implemented by organizations outside Hamamatsu. We do not have access to the data in these cookies, but we use these cookies to improve the overall website experience.
3. How do we use cookies?
This website uses cookies for following purposes:
- Certain cookies are necessary for our website to function. These are strictly necessary cookies and are required to enable website access, support navigation or provide relevant content. These cookies direct you to the correct region or country, and support security and ecommerce. Strictly necessary cookies also enforce your privacy preferences. Without these strictly necessary cookies, much of our website will not function.
- Analytics cookies are used to track website usage. This data enables us to improve our website usability, performance and website administration. In our analytics cookies, we do not store any personal identifying information.
- Functionality cookies. These are used to recognize you when you return to our website. This enables us to personalize our content for you, greet you by name and remember your preferences (for example, your choice of language or region).
- These cookies record your visit to our website, the pages you have visited and the links you have followed. We will use this information to make our website and the advertising displayed on it more relevant to your interests. We may also share this information with third parties for this purpose.
Cookies help us help you. Through the use of cookies, we learn what is important to our visitors and we develop and enhance website content and functionality to support your experience. Much of our website can be accessed if cookies are disabled, however certain website functions may not work. And, we believe your current and future visits will be enhanced if cookies are enabled.
4. Which cookies do we use?
There are two ways to manage cookie preferences.
- You can set your cookie preferences on your device or in your browser.
- You can set your cookie preferences at the website level.
If you don’t want to receive cookies, you can modify your browser so that it notifies you when cookies are sent to it or you can refuse cookies altogether. You can also delete cookies that have already been set.
If you wish to restrict or block web browser cookies which are set on your device then you can do this through your browser settings; the Help function within your browser should tell you how. Alternatively, you may wish to visit www.aboutcookies.org, which contains comprehensive information on how to do this on a wide variety of desktop browsers.
5. What are Internet tags and how do we use them with cookies?
Occasionally, we may use internet tags (also known as action tags, single-pixel GIFs, clear GIFs, invisible GIFs and 1-by-1 GIFs) at this site and may deploy these tags/cookies through a third-party advertising partner or a web analytical service partner which may be located and store the respective information (including your IP-address) in a foreign country. These tags/cookies are placed on both online advertisements that bring users to this site and on different pages of this site. We use this technology to measure the visitors' responses to our sites and the effectiveness of our advertising campaigns (including how many times a page is opened and which information is consulted) as well as to evaluate your use of this website. The third-party partner or the web analytical service partner may be able to collect data about visitors to our and other sites because of these internet tags/cookies, may compose reports regarding the website’s activity for us and may provide further services which are related to the use of the website and the internet. They may provide such information to other parties if there is a legal requirement that they do so, or if they hire the other parties to process information on their behalf.
If you would like more information about web tags and cookies associated with on-line advertising or to opt-out of third-party collection of this information, please visit the Network Advertising Initiative website http://www.networkadvertising.org.
6. Analytics and Advertisement Cookies
We use third-party cookies (such as Google Analytics) to track visitors on our website, to get reports about how visitors use the website and to inform, optimize and serve ads based on someone's past visits to our website.
You may opt-out of Google Analytics cookies by the websites provided by Google:
https://tools.google.com/dlpage/gaoptout?hl=en
As provided in this Privacy Policy (Article 5), you can learn more about opt-out cookies by the website provided by Network Advertising Initiative:
http://www.networkadvertising.org
We inform you that in such case you will not be able to wholly use all functions of our website.
Close