United Kingdom (EN)
Select your region or country.
An optogenetic tool for intracellular cAMP production
Since 2006, photo-sensitive ion channels have become an indispensable optogenetic tool in neuroscience. After forcibly expressing photo-sensitive ion channels on the specific neurons using genetic engineering techniques, these cells can be illuminated with a specific wavelength of light to excite or suppress each target neuron. Spatiotemporal accuracy of this technique is a feature not found in the conventional methods, such as pharmacological or electrical methods.
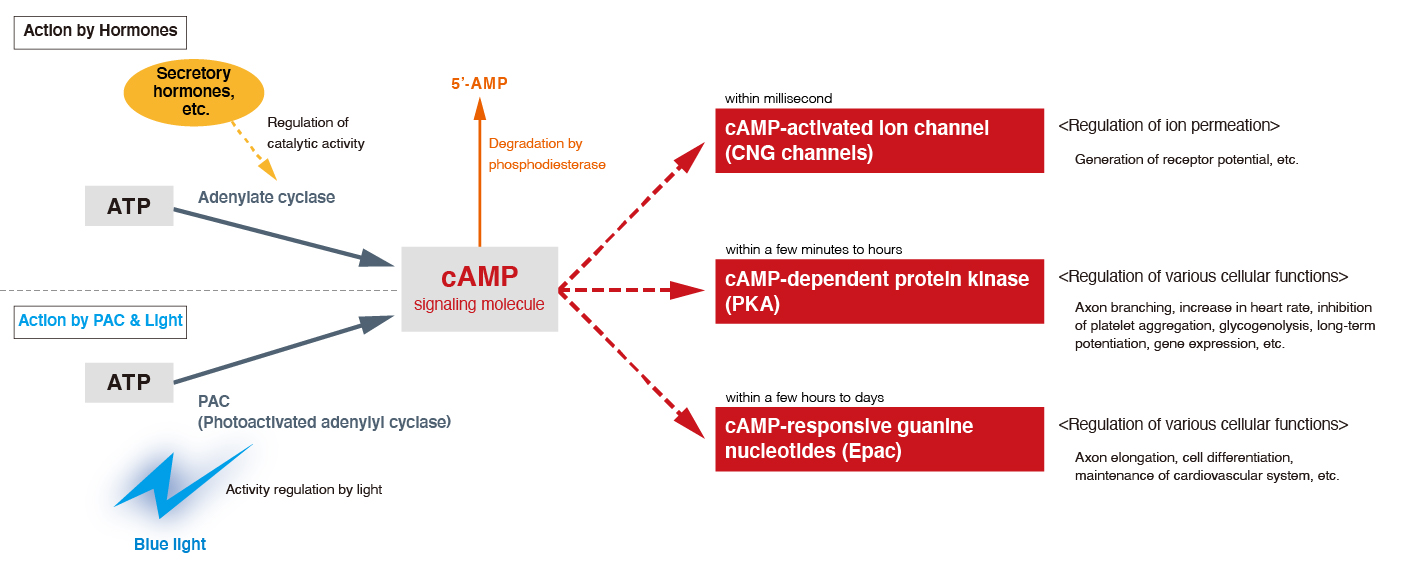
Incidentally, signaling factors, for instance, intracellular signal transduction pathways mediated by cyclic nucleotides such as cAMP and cGMP, are those that play essential roles for diverse and important biological functions. Activation of adenylate cyclase or inhibition of phosphodiesterase increases intracellular cAMP concentration and that’s what stimulates cyclic nucleotide-gated (CNG) channels, protein kinase A (PKA), and exchange protein directly activated by cAMP (Epac). These processes induce a variety of cellular responses, such as generation of receptor potentials, the axonal branching or elongation in neurons, increase in the heart rate, and glycogenolysis.
We will introduce an optogenetic tool "PAC" that can control cAMP production and induce these cellular responses triggered by various intensities of blue light.
■What is the optogenetics for controlling cell function via cAMP signaling pathway mediated by Photoactivated adenylyl cyclase (PAC)?
Function of PACs in microorganisms as a photosensor and its biomedical applications
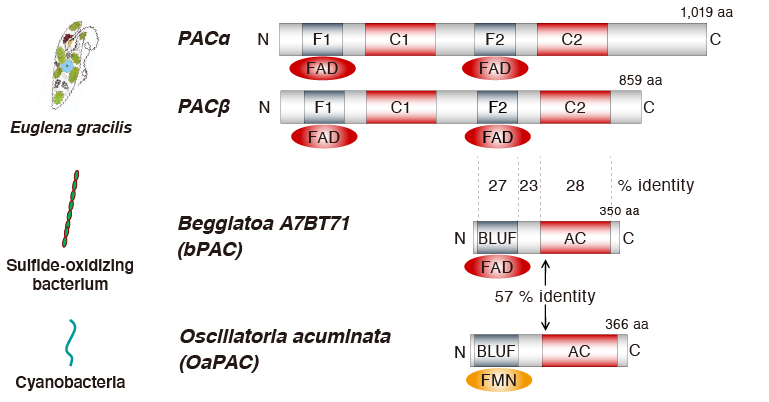
PAC was first identified in photosynthetic flagellate Euglena glacilis as photosensitive molecule inducing intracellular cAMP production by light illumination (Ref. 1). Recently, homologous proteins of PAC have been widely discovered in several prokaryotes.
Control of cAMP signaling by PACs is simple to handle because this process is independent of signal cascade mediated by G protein-coupled receptor GPCRs. Since PACs produce cAMP just by light illumination, cAMP production can be temporally and spatially regulated with high precision. Therefore, it is highly expected that this method enables to control various physiological functions inside cells and tissues directly by using only light illumination (Ref. 2).
■Scheme: PAC expression in cells and activation by blue light illumination
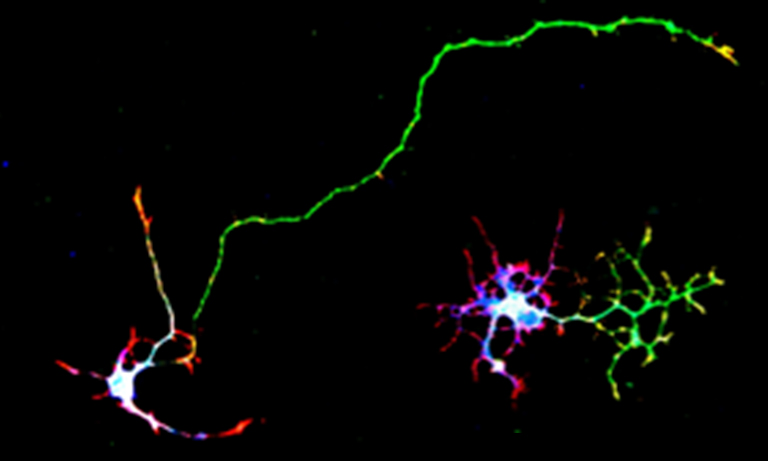
Excerpted from Scientific Reports 5, 19679, doi:10.1038/srep19679 (2016)
C-terminal deletion modification of OaPAC yields optogenetic tools with different photosensitivity
Among the PACs discovered so far, the structures of OaPAC from cyanobacteria and bPAC from sulfur bacteria have been determined. Both PACs have a light-sensitive BLUF domain at the N-terminus and an adenylate cyclase (AC) domain at the C-terminus, and these domains are arranged next to each other. Although the sequence and the structure of OaPAC and bPAC are similar, there is a hundred-fold difference in their photo-sensitivities (Ref. 3). This difference should provide a clue for modifying the design of PACs as an optogenetic tool applied to a lot of variations. In a collaboration with the Graduate School for the Creation of New Photonics Industries (GPI), we built lots of deletion mutants on the C-terminus and compared their enzymatic activities. We found that the longer the C-terminal region was ascribed the lower the enzymatic activity, and that the variation in their photoactivities was more than fifty-fold (Ref. 4).
■C-terminal amino acid sequence of various PACs

Amino acid sequence of OaPAC from Oscillatoria acuminata (NCBI, WP_015149803.1) and its deletion variants (Oa-363, Oa-360, Oa-357, Oa-354, Oa-351, Oa-348), bPAC from soil bacterium Beggiatoa (GenBank: GU461306.2), PACα from Euglena gracilis (GenBank: BAB85619.1) amino acid sequences in the C-terminal region. Yellow highlighting: conserved amino acids.
Excerpted from Scientific Reports 9, 20262 (2019)
User friendly PAC variants for researches
Low photosensitivity of wild-type OaPAC had been a bottleneck for practical use because of its susceptibility to phototoxicity due to strong light illumination. We have solved this problem and made it possible to use OaPACs for a wide range of applications. Two types of user-friendly OaPAC mutants with different photosensitivities, namely OaPAC360 and OaPAC348, are now commercially available as our products.
※These products are sold on consignment to COSMO BIO CO., LTD. For inquiries about the products, please visit the link below.
Click here for detailed product information (COSMO BIO CO., LTD. website)
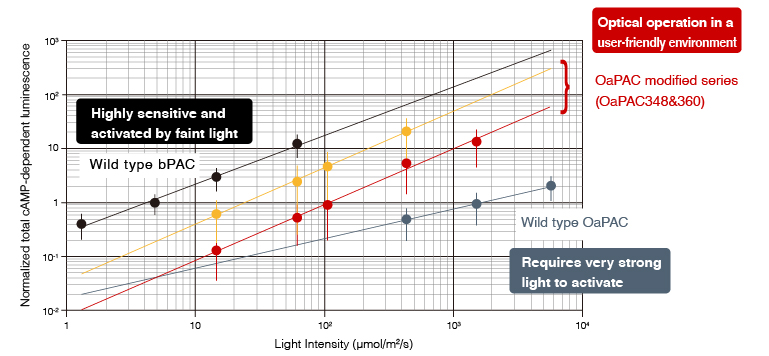
Excerpted from Scientific Reports 9, 20262 (2019)
References
- Iseki, M. et al. ”A blue-light-activated adenylyl cyclase mediates photoavoidance in Euglena gracilis. ”Nature 415, 1047-1051, doi:10.1038/4151047a (2002).
- Zhou, Z. et al. Photoactivated adenylyl cyclase (PAC) reveals novel mechanisms underlying cAMP-dependent axonal morphogenesis. Scientific Reports 5, 19679, doi:10.1038/srep19679 (2016).
- Ohki, M. et al. ”Structural insight into photoactivation of an adenylate cyclase from a photosynthetic cyanobacterium.” Proc Natl Acad Sci U S A 113, 6659-6664, doi:10.1073/pnas.1517520113 (2016).
- Hirano et al. “The C-terminal region affects the activity of photoactivated adenylyl cyclase from Oscillatoria acuminata.” Scientific Reports 9, Article number: 20262 (2019).
- Confirmation
-
It looks like you're in the . If this is not your location, please select the correct region or country below.
You're headed to Hamamatsu Photonics website for GB (English). If you want to view an other country's site, the optimized information will be provided by selecting options below.
In order to use this website comfortably, we use cookies. For cookie details please see our cookie policy.
- Cookie Policy
-
This website or its third-party tools use cookies, which are necessary to its functioning and required to achieve the purposes illustrated in this cookie policy. By closing the cookie warning banner, scrolling the page, clicking a link or continuing to browse otherwise, you agree to the use of cookies.
Hamamatsu uses cookies in order to enhance your experience on our website and ensure that our website functions.
You can visit this page at any time to learn more about cookies, get the most up to date information on how we use cookies and manage your cookie settings. We will not use cookies for any purpose other than the ones stated, but please note that we reserve the right to update our cookies.
1. What are cookies?
For modern websites to work according to visitor’s expectations, they need to collect certain basic information about visitors. To do this, a site will create small text files which are placed on visitor’s devices (computer or mobile) - these files are known as cookies when you access a website. Cookies are used in order to make websites function and work efficiently. Cookies are uniquely assigned to each visitor and can only be read by a web server in the domain that issued the cookie to the visitor. Cookies cannot be used to run programs or deliver viruses to a visitor’s device.
Cookies do various jobs which make the visitor’s experience of the internet much smoother and more interactive. For instance, cookies are used to remember the visitor’s preferences on sites they visit often, to remember language preference and to help navigate between pages more efficiently. Much, though not all, of the data collected is anonymous, though some of it is designed to detect browsing patterns and approximate geographical location to improve the visitor experience.
Certain type of cookies may require the data subject’s consent before storing them on the computer.
2. What are the different types of cookies?
This website uses two types of cookies:
- First party cookies. For our website, the first party cookies are controlled and maintained by Hamamatsu. No other parties have access to these cookies.
- Third party cookies. These cookies are implemented by organizations outside Hamamatsu. We do not have access to the data in these cookies, but we use these cookies to improve the overall website experience.
3. How do we use cookies?
This website uses cookies for following purposes:
- Certain cookies are necessary for our website to function. These are strictly necessary cookies and are required to enable website access, support navigation or provide relevant content. These cookies direct you to the correct region or country, and support security and ecommerce. Strictly necessary cookies also enforce your privacy preferences. Without these strictly necessary cookies, much of our website will not function.
- Analytics cookies are used to track website usage. This data enables us to improve our website usability, performance and website administration. In our analytics cookies, we do not store any personal identifying information.
- Functionality cookies. These are used to recognize you when you return to our website. This enables us to personalize our content for you, greet you by name and remember your preferences (for example, your choice of language or region).
- These cookies record your visit to our website, the pages you have visited and the links you have followed. We will use this information to make our website and the advertising displayed on it more relevant to your interests. We may also share this information with third parties for this purpose.
Cookies help us help you. Through the use of cookies, we learn what is important to our visitors and we develop and enhance website content and functionality to support your experience. Much of our website can be accessed if cookies are disabled, however certain website functions may not work. And, we believe your current and future visits will be enhanced if cookies are enabled.
4. Which cookies do we use?
There are two ways to manage cookie preferences.
- You can set your cookie preferences on your device or in your browser.
- You can set your cookie preferences at the website level.
If you don’t want to receive cookies, you can modify your browser so that it notifies you when cookies are sent to it or you can refuse cookies altogether. You can also delete cookies that have already been set.
If you wish to restrict or block web browser cookies which are set on your device then you can do this through your browser settings; the Help function within your browser should tell you how. Alternatively, you may wish to visit www.aboutcookies.org, which contains comprehensive information on how to do this on a wide variety of desktop browsers.
5. What are Internet tags and how do we use them with cookies?
Occasionally, we may use internet tags (also known as action tags, single-pixel GIFs, clear GIFs, invisible GIFs and 1-by-1 GIFs) at this site and may deploy these tags/cookies through a third-party advertising partner or a web analytical service partner which may be located and store the respective information (including your IP-address) in a foreign country. These tags/cookies are placed on both online advertisements that bring users to this site and on different pages of this site. We use this technology to measure the visitors' responses to our sites and the effectiveness of our advertising campaigns (including how many times a page is opened and which information is consulted) as well as to evaluate your use of this website. The third-party partner or the web analytical service partner may be able to collect data about visitors to our and other sites because of these internet tags/cookies, may compose reports regarding the website’s activity for us and may provide further services which are related to the use of the website and the internet. They may provide such information to other parties if there is a legal requirement that they do so, or if they hire the other parties to process information on their behalf.
If you would like more information about web tags and cookies associated with on-line advertising or to opt-out of third-party collection of this information, please visit the Network Advertising Initiative website http://www.networkadvertising.org.
6. Analytics and Advertisement Cookies
We use third-party cookies (such as Google Analytics) to track visitors on our website, to get reports about how visitors use the website and to inform, optimize and serve ads based on someone's past visits to our website.
You may opt-out of Google Analytics cookies by the websites provided by Google:
https://tools.google.com/dlpage/gaoptout?hl=en
As provided in this Privacy Policy (Article 5), you can learn more about opt-out cookies by the website provided by Network Advertising Initiative:
http://www.networkadvertising.org
We inform you that in such case you will not be able to wholly use all functions of our website.
Close


























































































































