United Kingdom (EN)
Select your region or country.
Quantaurus-QY® Absolute PL quantum yield spectrometer
C11347 series
The Quantaurus-QY is a compact, easy-to-use instrument for measuring the quantum yield of photoluminescent materials. It performs absolute measurements and does not require known reference standards, as typically required when using the traditional relative method. Samples in different forms -thin films, solids, powders, and solutions- can be analyzed. Liquid samples can be cooled down to -196 deg.C (77K) with liquid nitrogen.
Quantaurus-QY is a registered trademark of Hamamatsu Photonics K.K. (China, EU, Japan, Korea, Taiwan, UK, USA).
Features
Instantaneous measurement
The multichannel detector captures the sensitivity-compensated spectrum, and calculates the quantum yield in a process that instantaneously finds the absolute value of the quantum yield.
Dialog-style dedicated software keeps the measurement process simple.
Fully automated hardware
The software-controlled monochromator allows selecting excitation wavelengths so that the sample can be excited by various excitation wavelengths. Wavelength dependence of quantum yields and excitation spectrum can be automatically measured.
Analyzing different sample forms
Quantaurus-QY handles solution, thin-film, and powder samples. With a Dewar flask holder, solution samples can be cooled by liquid nitrogen to -196 ˚C (77 K).

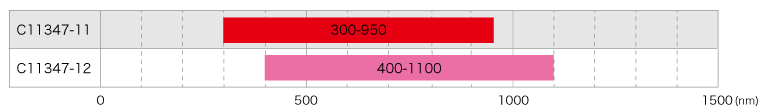
2 models available
Two product types are provided according to the wavelength range for sample excitation and photoluminescence: one covers a spectral range from 300 nm to 950 nm and the other from 400 nm to 1100 nm.
Applications
- Organic metal complex
- Fluorescent probe
- Dye sensitized PV material
- OLED material
- Quantum dot
- LED phosphor
Principle of quantum yield measurement
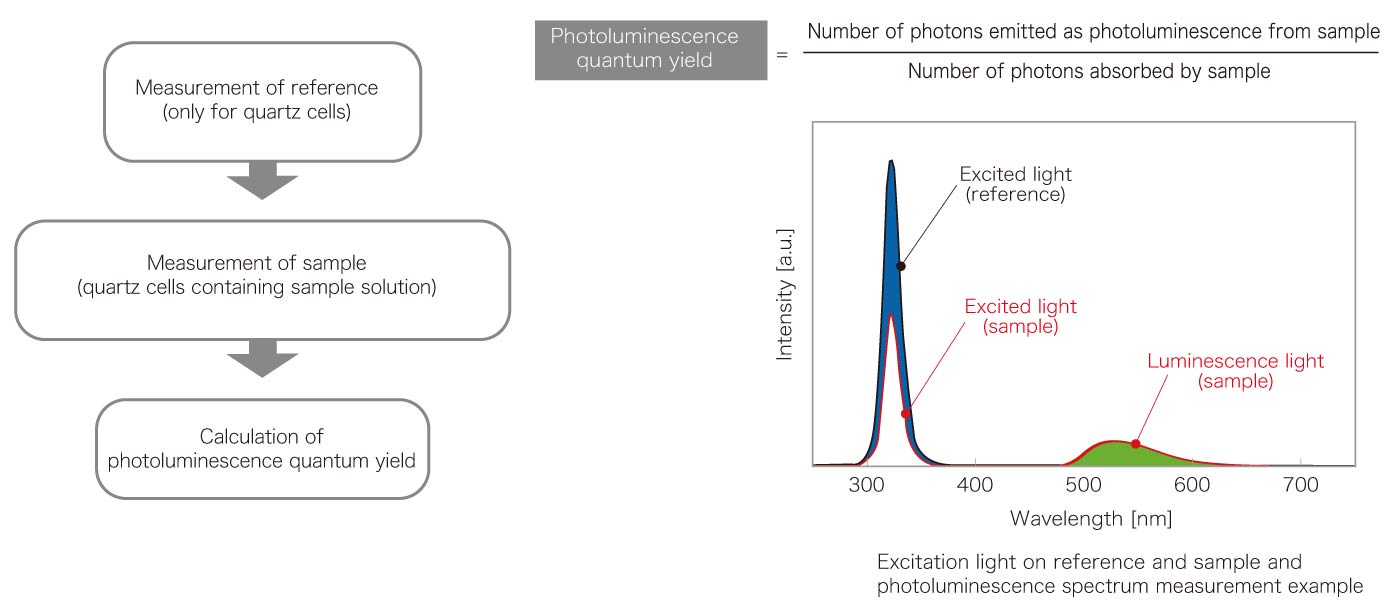
Quantaurus-QY operation instruction
Quantaurus-QY is a compact instrument that integrates an excitation light source, detector, and optical system into a single unit, eliminating the need for researchers to adjust many components that previously required long time and labor for measurement. By simply loading a sample and giving a few instructions, luminescence quantum yield, excitation wavelength dependence, PL excitation spectrum, etc. are measured in a short time.
Specifications
| Type number | C11347-11 | C11347-12 |
|---|---|---|
| PL measurement wavelength range | 300 nm to 950 nm | 400nm to 1100 nm |
| Monochromatic light source | ||
| Light source | 150 W xenon light source | |
| Excitation wavelength | 250 nm to 850 nm | 375 nm to 850 nm |
| Bandwidth | 10 nm or less (FWHM) | |
| Excitation wavelength control | Automatic control | |
| Multichannel spectroscope | ||
| Measurement wavelength range | 200 nm to 950 nm | 350 nm to 1100 nm |
| Wavelength resolution | < 2 nm | < 2.5 nm |
| Number of photosensitive device channels | 1024ch | |
| Device cooling temperature | - 15 ℃ | |
| AD resolution | 16 bit | |
| Spectroscope optical arrangement | Czerny-Turnre type | |
| Integrating sphere | ||
| Material | Spectralon | |
| Size | 3.3 inch | |
| Software | ||
| Measurement items | PL quantum yield | |
| Excitation wavelength dependence of quantum yield | ||
| PL spectrum (peak wavelength, FWHM) | ||
| PL excitation spectrum | ||
| Color measurement (chromaticity, color temperature, color rendering index, etc.) | ||
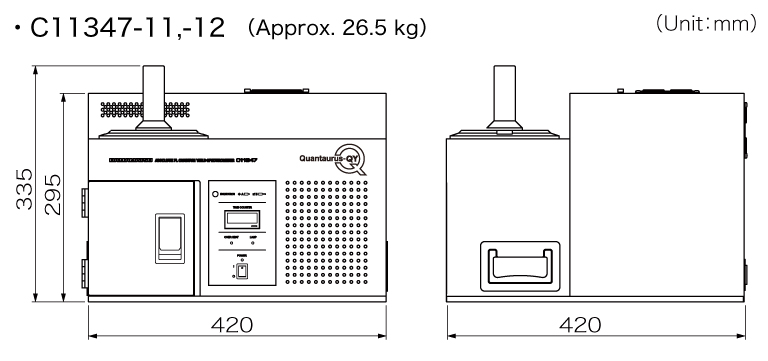
Dimensions
Publications
| Authors | Title | Journal name | Volume Page | Page | Year |
|---|---|---|---|---|---|
| A. Endo, K. Suzuki, T. Yoshihara, S. Tobita, M. Yahiro. and C. Adachi | Measurement of photoluminescence efficiency of Ir(III) phenylpyridine derivatives in solution and solid-state films | Chem. Phy. Lett. | 460 | 155 | 2008 |
| T. Sajoto, P. I. Djurovich, A. B. Tamayo, J. Oxgaard, W. A. Goddard III, and M. E. Thompson | Temperature Dependence of Blue Phosphorescent Cyclometalated Ir(III) Complexes | J. Am. Chem. Soc. | 131 | 9813 | 2009 |
| H.-F. Chen, S.-J. Yang, Z.-H. Tsai, W.-Y. Hung, T.-C. Wang, and K.-T. Wong | 1,3,5-Triazine derivatives as new electron transport–type host materials for highly efficient green phosphorescent OLEDs | J. Mater. Chem. | 19 | 8112 | 2009 |
| H. J. Bolink, L. Cappelli, S. Cheylan, E. Coronado, R. D. Costa, N. Lardies, Md. K. Nazeeruddin, and E. Orti | Origin of the large spectral shift in electroluminescence in a blue light emitting cationic iridium(III) complex | J. Mater. Chem. | 17 | 5032 | 2007 |
| R. D. Costa, F. J. Cespedes-Guirao, H. J. Bolink, F. Fernandez-Lazaro, A. Sastre-Santos, E. Orti, and J. Gierschner | A Deep-Red-Emitting Perylenediimide-Iridium-Complex Dyad: Following the Photophysical Deactivation Pathways | J. Phys. Chem. C | 113 | 19292 | 2009 |
| R. D. Costa, F. Fernandez, L. Sanchez, N. Martin, E. Orti, and H. J. Bolink | Dumbbell-Shaped Dinuclear Iridium Complexes and Their Application to Light-Emitting Electrochemical Cells | Chem. Eur. J | 16 | 9855 | 2010 |
| R. D. Costa, E. Orti, H. J. Bolink, S. Graber, C. E. Housecroft, and E. C. Constable | Efficient and Long-Living Light-Emitting Electrochemical Cells | Adv. Funct. Mater. | 20 | 1511 | 2010 |
| R. D. Costa, E. Orti, D. Tordera, H. J. Bolink, S. Graber, C. E. Housecroft, L. Sachno, M. Neuburger, and E. C. Constable | Stable and Efficient Solid-State Light-Emitting Electrochemical Cells Based on a Series of Hydrophobic Iridium Complexes | Adv. Funct. Mater. | 1 | 282 | 2011 |
| Authors | Title | Journal name | Volume | Page | Year |
|---|---|---|---|---|---|
| T. Nakajima, M. Isobe, T. Tsuchiya, Y. Ueda, and T. Kumagai | Direct fabrication of metavanadate phosphor films on organic substrates for white-light-emitting devices | Nature Materials | 7 | 735 | 2008 |
| T. Ogi, Y. Kaihatsu, F. Iskandar, W.-N. Wang, and K. Okuyama | Facile Sunthesis of New Full-Color-Emitting BCNO Phosphors with High Quantum Efficiency | Adv. Mater | 20 | 3235 | 2008 |
| Authors | Title | Journal name | Volume Page | Page | Year |
|---|---|---|---|---|---|
| H. Ito, M. Matsuoka, Y. Ueda, M. Takuma, Y. Kudo, and K. Iguchi | Quinolinecarboxylic acid based fluorescent molecules: ratiometric response to Zn2+ | Tetrahedron | 65 | 4235 | 2009 |
| S. Kamino, H. Ichihara, S. Wada, Y. Horio, Y. Usami, T. Yamaguchi, T. Koda, A. Harada, K. Shimanuki, M. Arimoto, M. Doi, and Y. Fujita | Design and synthesis of regioisomerically pure unsymmetrical xanthene derivatives for staining live cells and their photochemical properties | Bioorg. Med. Chem. Lett. | 18 | 4380 | 2008 |
| Y. Mikata, A. Yamashita, A. Kawamura, H. Konno, Y. Miyamoto, and S. Tamotsu | Bisquinoline-based fluorescent zinc sensors | Dalton Trans. | 3800 | 2009 | |
| Takahisa Suzuki, Seisuke Arai, Mayumi Takeuchi, Chiye Sakurai, Hideaki Ebana, Tsunehito Higashi, Hitoshi Hashimoto, Kiyotaka Hatsuzawa, Ikuo Wada | Development of Cysteine-Free Fluorescent Proteins for the Oxidative Environment | PLoS ONE | 7 | e37551 | 2012 |
| Authors | Title | Journal name | Volume Page | Page | Year |
|---|---|---|---|---|---|
| A. Ishii, K. Habu, S. Kishi, H. Otsu, T. Komatsu, K. Osaka, K. Kato, S. Kimura, M. Tanaka, M. Hasegawa, and Y. Shigesato | Novel emission properties of melem caused by the heavy metal effect of lanthanides(III) in a LB film | Photochem. Photobiol. Sci. | 6 | 804 | 2007 |
| K. Matsumoto, N. Matsumoto, A. Ishii, T. Tsukuda, M. Hasegawa, and T. Tsubomura | Structural and spectroscopic properties of a copper(I)–bis(N-heterocyclic)carbene complex | Dalton Trans. | 6795 | 2009 | |
| Y. Matano, T. Miyajima, N. Ochi, Y. Nakao, S. Sakai, and H. Imahori | Synthesis of Thiophene-Containing Hybrid Calixphyrins of the 5,10-Porphodimethene Type | J. Org. Chem. | 73 (13) | 5139 | 2008 |
| D. Kuzuhara, J. Mack, H. Yamada, T. Okujima, N. Ono, and N. Kobayashi | Synthesis, Structures, and Optical and Electrochemical Properties of Benzoporphycenes | Chem. Eur. J | 15 | 10060 | 2009 |
| D. Maeda, H. Shimakoshi, M. Abe, M. Fujitsuka, T. Majima, and Y. Hisaeda | Synthesis of a Novel Sn)IV) Porphycene-Ferrocene Triad Linked by Axal Coordination and Solvent Polarity Effect in Photoinduced Charge Separation Process | Inorg. Chem. | 49 | 2872 | 2010 |
| D. Maeda, H. Shimakoshi, M. Abe, and Y. Hisaeda | Syntheses and Photophysical Behavior of Porphyrin Isomer Sn(IV) Complexes | Inorg. Chem. | 48 | 9853 | 2009 |
| H. Shimakoshi, T. Baba, Y. iseki, I. Aritome, A. Endo, C. Adachi, and Y. Hisaeda | Photophysical and photosensitizing properties of brominated porphycenes | Chem. Commun. | 2882 | 2008 |
Case study
To evaluate the organic optoelectronic materials and devices being developed by the Center for Organic Photonics and Electronics Research (OPERA), Kyushu University, various measurement methods such as photoluminescence(PL) quantum yield measurement and fluorescence lifetime measurement are required. For these evaluations, our Quantaurus series and streak cameras have been introduced.
We interviewed Prof. Chihaya Adachi, Director of the Center, and Assoc. Prof. Hajime Nakanotani about establishing absolute photoluminescence quantum yield measurement methods, the impact of our Quantaurus series on their research, and their prospects for future research.
Applications : Evaluation of luminescent materials
- Confirmation
-
It looks like you're in the . If this is not your location, please select the correct region or country below.
You're headed to Hamamatsu Photonics website for GB (English). If you want to view an other country's site, the optimized information will be provided by selecting options below.
In order to use this website comfortably, we use cookies. For cookie details please see our cookie policy.
- Cookie Policy
-
This website or its third-party tools use cookies, which are necessary to its functioning and required to achieve the purposes illustrated in this cookie policy. By closing the cookie warning banner, scrolling the page, clicking a link or continuing to browse otherwise, you agree to the use of cookies.
Hamamatsu uses cookies in order to enhance your experience on our website and ensure that our website functions.
You can visit this page at any time to learn more about cookies, get the most up to date information on how we use cookies and manage your cookie settings. We will not use cookies for any purpose other than the ones stated, but please note that we reserve the right to update our cookies.
1. What are cookies?
For modern websites to work according to visitor’s expectations, they need to collect certain basic information about visitors. To do this, a site will create small text files which are placed on visitor’s devices (computer or mobile) - these files are known as cookies when you access a website. Cookies are used in order to make websites function and work efficiently. Cookies are uniquely assigned to each visitor and can only be read by a web server in the domain that issued the cookie to the visitor. Cookies cannot be used to run programs or deliver viruses to a visitor’s device.
Cookies do various jobs which make the visitor’s experience of the internet much smoother and more interactive. For instance, cookies are used to remember the visitor’s preferences on sites they visit often, to remember language preference and to help navigate between pages more efficiently. Much, though not all, of the data collected is anonymous, though some of it is designed to detect browsing patterns and approximate geographical location to improve the visitor experience.
Certain type of cookies may require the data subject’s consent before storing them on the computer.
2. What are the different types of cookies?
This website uses two types of cookies:
- First party cookies. For our website, the first party cookies are controlled and maintained by Hamamatsu. No other parties have access to these cookies.
- Third party cookies. These cookies are implemented by organizations outside Hamamatsu. We do not have access to the data in these cookies, but we use these cookies to improve the overall website experience.
3. How do we use cookies?
This website uses cookies for following purposes:
- Certain cookies are necessary for our website to function. These are strictly necessary cookies and are required to enable website access, support navigation or provide relevant content. These cookies direct you to the correct region or country, and support security and ecommerce. Strictly necessary cookies also enforce your privacy preferences. Without these strictly necessary cookies, much of our website will not function.
- Analytics cookies are used to track website usage. This data enables us to improve our website usability, performance and website administration. In our analytics cookies, we do not store any personal identifying information.
- Functionality cookies. These are used to recognize you when you return to our website. This enables us to personalize our content for you, greet you by name and remember your preferences (for example, your choice of language or region).
- These cookies record your visit to our website, the pages you have visited and the links you have followed. We will use this information to make our website and the advertising displayed on it more relevant to your interests. We may also share this information with third parties for this purpose.
Cookies help us help you. Through the use of cookies, we learn what is important to our visitors and we develop and enhance website content and functionality to support your experience. Much of our website can be accessed if cookies are disabled, however certain website functions may not work. And, we believe your current and future visits will be enhanced if cookies are enabled.
4. Which cookies do we use?
There are two ways to manage cookie preferences.
- You can set your cookie preferences on your device or in your browser.
- You can set your cookie preferences at the website level.
If you don’t want to receive cookies, you can modify your browser so that it notifies you when cookies are sent to it or you can refuse cookies altogether. You can also delete cookies that have already been set.
If you wish to restrict or block web browser cookies which are set on your device then you can do this through your browser settings; the Help function within your browser should tell you how. Alternatively, you may wish to visit www.aboutcookies.org, which contains comprehensive information on how to do this on a wide variety of desktop browsers.
5. What are Internet tags and how do we use them with cookies?
Occasionally, we may use internet tags (also known as action tags, single-pixel GIFs, clear GIFs, invisible GIFs and 1-by-1 GIFs) at this site and may deploy these tags/cookies through a third-party advertising partner or a web analytical service partner which may be located and store the respective information (including your IP-address) in a foreign country. These tags/cookies are placed on both online advertisements that bring users to this site and on different pages of this site. We use this technology to measure the visitors' responses to our sites and the effectiveness of our advertising campaigns (including how many times a page is opened and which information is consulted) as well as to evaluate your use of this website. The third-party partner or the web analytical service partner may be able to collect data about visitors to our and other sites because of these internet tags/cookies, may compose reports regarding the website’s activity for us and may provide further services which are related to the use of the website and the internet. They may provide such information to other parties if there is a legal requirement that they do so, or if they hire the other parties to process information on their behalf.
If you would like more information about web tags and cookies associated with on-line advertising or to opt-out of third-party collection of this information, please visit the Network Advertising Initiative website http://www.networkadvertising.org.
6. Analytics and Advertisement Cookies
We use third-party cookies (such as Google Analytics) to track visitors on our website, to get reports about how visitors use the website and to inform, optimize and serve ads based on someone's past visits to our website.
You may opt-out of Google Analytics cookies by the websites provided by Google:
https://tools.google.com/dlpage/gaoptout?hl=en
As provided in this Privacy Policy (Article 5), you can learn more about opt-out cookies by the website provided by Network Advertising Initiative:
http://www.networkadvertising.org
We inform you that in such case you will not be able to wholly use all functions of our website.
Close





























































































































